Surfactant
Surfactants are compounds that lower the
surface tension of a liquid, the interfacial tension between two liquids, or that between a liquid and a solid. Surfactants may act as
detergents,
wetting agents,
emulsifiers,
foaming agents, and
dispersants.

 Micelle
Micelle in aqueous solution
Etymology and definition
The term
surfactant/surfactants is a
blend of
surface active agents.[SUP]
[1][/SUP]
In
Index Medicus and the
United States National Library of Medicine,
surfactant/surfactants is reserved for the meaning
pulmonary surfactant. For the more general meaning,
surface active agent/s is the heading.
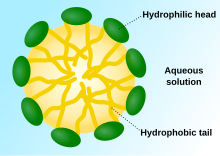

A
micelle—the
lipophilic tails of the surfactant ions remain on the inside of the micelle due to unfavourable interactions. The polar "heads" of the micelle, due to favourable interactions with water, form a
hydrophilic outer layer that in effect protects the hydrophobic core of the micelle. The compounds that make up a micelle are typically amphiphilic in nature, meaning that micelles are soluble not only in protic solvents such as water but also in aprotic solvents as a reverse micelle.
Composition and structure
Surfactants are usually
organic compounds that are
amphiphilic, meaning they contain both
hydrophobic groups (their
tails) and
hydrophilic groups (their
heads).[SUP]
[2][/SUP] Therefore, a surfactant contains both a water insoluble (or oil soluble) component and a water soluble component. Surfactants will diffuse in water and
adsorb at interfaces between air and water or at the interface between oil and water, in the case where water is mixed with oil. The insoluble hydrophobic group may extend out of the bulk water phase, into the air or into the oil phase, while the water soluble head group remains in the water phase. This alignment of surfactants at the surface modifies the surface properties of water at the water/air or water/oil interface.
World production of surfactants is estimated at 15 Mton/y, of which about half are soaps. Other surfactants produced on a particularly large scale are
linear alkylbenzenesulfonates (1700 kton/y),
lignin sulfonates (600 kton/y),
fatty alcohol ethoxylates (700 ktons/y),
alkylphenol ethoxylates (500 kton/y).[SUP]
[3][/SUP]
-

Sodium stearate, the most common component of most soap, which comprise about 50% of commercial surfactants.
-

4-(5-Dodecyl) benzenesulfonate, a linear dodecylbenzenesulfonate, one of the most common surfactants.
Structure of surfactant phases in water
In the bulk aqueous phase, surfactants form aggregates, such as
micelles, where the hydrophobic tails form the core of the aggregate and the hydrophilic heads are in contact with the surrounding liquid. Other types of aggregates such as spherical or cylindrical micelles or bilayers can be formed. The shape of the aggregates depends on the chemical structure of the surfactants, depending on the balance of the sizes of the hydrophobic tail and hydrophilic head. This is known as the HLB, Hydrophilic-lipophilic balance. Surfactants reduce the
surface tension of water by
adsorbing at the liquid-gas interface. The relation that links the surface tension and the surface excess is known as the
Gibbs isotherm.
Dynamics of surfactants at interfaces
The dynamics of adsorption of surfactants is of great importance for practical applications such as foaming, emulsifying or coating processes, where bubbles or drops are rapidly generated and need to be stabilized. The dynamics of adsorption depends on the diffusion coefficient of the surfactants. Indeed, as the interface is created, the adsorption is limited by the diffusion of the surfactants to the interface. In some cases, there exists a barrier of energy for the adsorption or the desorption of the surfactants, then the adsorption dynamics is known as ‘kinetically limited'. Such energy barrier can be due to steric or electrostatic repulsions. The
surface rheology of surfactant layers, including the elasticity and viscosity of the surfactant layers plays a very important role in foam or emulsion stability.
Characterization of interfaces and surfactant layers
Interfacial and surface tension can be characterized by classical methods such as the -pendant or spinning drop method Dynamic surface tensions, i.e. surface tension as a function of time, can be obtained by the
Maximum Bubble Pressure apparatus
The structure of surfactant layers can be studied by
ellipsometry or X-Ray reflectivity.
Surface rheology can be characterized by the oscillating drop method or shear surface rheometers such as double-cone, double-ring or magnetic rod shear surface rheometer.
Detergents in biochemistry and biotechnology
In solution, detergents help solubilize a variety of chemical species by dissociating aggregates and unfolding proteins. Popular surfactants in the biochemistry laboratory are
SDS and
CTAB. Detergents are key reagents to
extract protein by lysis of the cells and tissues: They disorganize the membrane's lipidic bilayer (SDS,
Triton X-100,
X-114,
CHAPS,
DOC, and
NP-40), and solubilize proteins. Milder detergents such as (OctylThioGlucosides) are used to solubilize sensible proteins (
enzymes,
receptors). Non-solubilized material is harvested by centrifugation or other means. For
electrophoresis, for example, proteins are classically treated with
SDS to denature the native
tertiary and quaternary structures, allowing the separation of proteins according to their
molecular weight.
Detergents have also been used to decellularise organs. This process maintains a matrix of proteins that preserves the structure of the organ and often the microvascular network. The process has been successfully used to prepare organs such as the liver and heart for transplant in rats.[SUP]
[4][/SUP]
Pulmonary surfactants are also naturally secreted by type II cells of the lung
alveoli in
mammals.
Classification of surfactants
The "tail" of most surfactants are fairly similar, consisting of a
hydrocarbon chain, which can be branch, linear, or aromatic.
Fluorosurfactants have
fluorocarbon chains.
Siloxane surfactants have
siloxane chains.
Many important surfactants include a polyether chain terminating in a highly polar anionic group. The polyether groups often comprise ethoxylated (
polyethylene oxide-like) sequences inserted to increase the hydrophilic character of a surfactant.
Polypropylene oxides conversely, may be inserted to increase the lipophilic character of a surfactant.
Surfactant molecules have either one tail or two; those with two tails are said to be
double-chained.


Surfactant classification according to the composition of their head: nonionic, anionic, cationic, amphoteric.
Most commonly, surfactants are classified according to polar head group. A non-ionic surfactant has no charge groups in its head. The head of an ionic surfactant carries a net charge. If the charge is negative, the surfactant is more specifically called anionic; if the charge is positive, it is called cationic. If a surfactant contains a head with two oppositely charged groups, it is termed zwitterionic. Commonly encountered surfactants of each type include:
Anionic
Sulfate, sulfonate, and phosphate esters
Anionic surfactants contain anionic functional groups at their head, such as
sulfate,
sulfonate,
phosphate, and
carboxylates. Prominent alkyl sulfates include
ammonium lauryl sulfate,
sodium lauryl sulfate (SDS, sodium dodecyl sulfate, another name for the compound) and the related alkyl-ether sulfates
sodium laureth sulfate, also known as sodium lauryl ether sulfate (SLES), and
sodium myreth sulfate.
Docusates:
dioctyl sodium sulfosuccinate,
perfluorooctanesulfonate (PFOS),
perfluorobutanesulfonate,
linear alkylbenzene sulfonates (LABs).
These include alkyl-aryl ether phosphates and the alkyl ether phosphate
Carboxylates
These are the most common surfactants and comprise the alkyl carboxylates (soaps), such as
sodium stearate. More specialized species include
sodium lauroyl sarcosinate and carboxylate-based fluorosurfactants such as
perfluorononanoate,
perfluorooctanoate (PFOA or PFO).
Cationic head groups
- pH-dependent primary, secondary, or tertiary amines: Primary amines become positively charged at pH < 10, secondary amines become charged at pH < 4:
- Permanently charged quaternary ammonium cation:
Zwitterionic surfactants
Zwitterionic (
amphoteric) surfactants have both cationic and anionic centers attached to the same molecule. The cationic part is based on primary, secondary, or tertiary
amines or quaternary ammonium cations. The anionic part can be more variable and include sulfonates, as in
CHAPS (3-[(3-Cholamidopropyl)dimethylammonio]-1-propanesulfonate). Other anionic groups are
sultaines illustrated by
cocamidopropyl hydroxysultaine.
Betaines, e.g.,
cocamidopropyl betaine. Phosphates:
lecithin
Nonionic surfactant
Many long chain alcohols exhibit some surfactant properties. Prominent among these are the
fatty alcohols cetyl alcohol,
stearyl alcohol, and
cetostearyl alcohol (consisting predominantly of cetyl and stearyl alcohols), and
oleyl alcohol.
According to the composition of their counter-ion
In the case of ionic surfactants, the counter-ion can be:
Current market and forecast
The annual global production of surfactants was 13 million metric tons in 2008, and the annual turnover reached US$24.33 billion in 2009, nearly 2% up from the previous year. The market is expected to experience quite healthy growth by 2.8% annually to 2012 and by 3.5 – 4% thereafter.[SUP]
[5][/SUP][SUP]
[6][/SUP] Specialists expect the global surfactant market to generate revenues of more than US$41 billion in 2018 – translating to an average annual growth of 4.5%[SUP]
[7][/SUP]
Health and environmental controversy
Surfactants are routinely deposited in numerous ways on land and into water systems, whether as part of an intended process or as industrial and household waste. Some of them are known to be toxic to animals, ecosystems, and humans, and can increase the diffusion of other environmental contaminants.[SUP]
[8][/SUP][SUP]
[9][/SUP][SUP]
[10][/SUP] As a result, there are proposed or voluntary restrictions on the use of some surfactants. For example,
PFOS is a
persistent organic pollutant as judged by the
Stockholm Convention. Additionally,
PFOA has been subject to a voluntary agreement by the
U.S. Environmental Protection Agency and eight chemical companies to reduce and eliminate emissions of the chemical and its precursors.[SUP]
[11][/SUP]
The two major surfactants used in the year 2000 were linear alkylbenzene
sulfonates (LAS) and the alkyl phenol
ethoxylates (APE). They break down in the
aerobic conditions found in
sewage treatment plants and in soil.[SUP]
[12][/SUP]
Ordinary dishwashing
detergent, for example, will promote water penetration in soil, but the effect would last only a few days (many standard laundry detergent powders contain levels of chemicals such as
alkali and
chelating agents that can be damaging to plants and should not be applied to soils). Commercial soil wetting agents will continue to work for a considerable period, but they will eventually be degraded by soil micro-organisms. Some can, however, interfere with the life-cycles of some aquatic organisms, so care should be taken to prevent run-off of these products into streams, and excess product should not be washed down.[SUP]
[citation needed][/SUP]
Anionic surfactants can be found in soils as the result of sludge application, wastewater irrigation, and remediation processes. Relatively high concentrations of surfactants together with multimetals can represent an environmental risk. At low concentrations, surfactant application is unlikely to have a significant effect on trace metal mobility.[SUP]
[13][/SUP][SUP]
[14][/SUP]
Biosurfactants
Biosurfactants are surface-active substances synthesised by living cells.[SUP]
[citation needed][/SUP] Interest in microbial surfactants has been steadily increasing in recent years due to their diversity, environmentally friendly nature, possibility of large-scale production, selectivity, performance under extreme conditions, and potential applications in environmental protection.[SUP]
[15][/SUP][SUP]
[16][/SUP]
Biosurfactants enhance the emulsification of hydrocarbons, have the potential to solubilise hydrocarbon contaminants and increase their availability for microbial degradation. The use of chemicals for the treatment of a hydrocarbon polluted site may contaminate the environment with their by-products, whereas biological treatment may efficiently destroy pollutants, while being biodegradable themselves. Hence, biosurfactant-producing microorganisms may play an important role in the accelerated bioremediation of hydrocarbon-contaminated sites.[SUP]
[17][/SUP][SUP]
[18][/SUP][SUP]
[19][/SUP] These compounds can also be used in
enhanced oil recovery and may be considered for other potential applications in environmental protection.[SUP]
[19][/SUP][SUP]
[20][/SUP] Other applications include herbicides and pesticides formulations, detergents, healthcare and cosmetics, pulp and paper, coal, textiles, ceramic processing and food industries, uranium ore-processing, and mechanical dewatering of peat.[SUP]
[15][/SUP][SUP]
[16][/SUP][SUP]
[21][/SUP]
Several microorganisms are known to synthesise surface-active agents; most of them are bacteria and yeasts.[SUP]
[22][/SUP][SUP]
[23][/SUP] When grown on hydrocarbon substrate as the carbon source, these microorganisms synthesise a wide range of chemicals with surface activity, such as glycolipid, phospholipid, and others.[SUP]
[24][/SUP][SUP]
[25][/SUP] These chemicals are synthesised to emulsify the hydrocarbon substrate and facilitate its transport into the cells. In some bacterial species such as
Pseudomonas aeruginosa, biosurfactants are also involved in a group motility behavior called
swarming motility.
Safety and environmental risks
Most anionic and nonionic surfactants are nontoxic, having
LD50 comparable to sodium chloride. The situation for cationic surfactants is more diverse. Dialkyldimethylammonium chlorides have very low
LD50's (5 g/kg) but alkylbenzyldimethylammonium chloride has an LD50 of 0.35 g/kg. Prolonged exposure of skin to surfactants can cause chaffing because surfactants (e.g., soap) disrupts the lipid coating that protects skin (and other) cells.[SUP]
[3][/SUP]
Biosurfactants and Deepwater Horizon
The use of biosurfactants as a way to remove
petroleum from contaminated sites has been questioned, and criticized as environmentally unsafe. Biosurfactants were not used by
BP after the
Deepwater Horizon oil spill. However, unprecedented amounts of
Corexit (active ingredient:
Tween-80), were sprayed directly into the ocean at the leak and on the sea-water's surface, the theory being that the surfactants isolate droplets of oil, making it easier for petroleum-consuming microbes to digest the oil.
Applications
Surfactants play an important role as cleaning,
wetting,
dispersing,
emulsifying,
foaming and
anti-foaming agents in many practical applications and products, including:
See also







